HIV cure research: advances and prospects
HAL Id: pasteur-01420527
Published Time: Thu, 19 Mar 2026 17:22:51 GMT
Number of Pages: 40
https://www.carmenthyssenmalaga.org on 24 Apr 2017
HAL is a multi-disciplinary open access archive for the deposit and dissemination of scientific research documents, whether they are published or not. The documents may come from teaching and research institutions in France or abroad, or from public or private research centers. L’archive ouverte pluridisciplinaire HAL, est destinée au dépôt et à la diffusion de documents scientifiques de niveau recherche, publiés ou non, émanant des établissements d’enseignement et de recherche français ou étrangers, des laboratoires publics ou privés.
Distributed under a Creative Commons CC BY-NC-SA 4.0 - Attribution - Non-commercial use - ShareAlike - International License
HIV cure research: advances and prospects.
Caroline P Passaes, Asier Sáez-Cirión
To cite this version:
Caroline P Passaes, Asier Sáez-Cirión. HIV cure research: advances and prospects.. Virology, 2014, 454-455, pp.340-52. ⟨10.1016/j.virol.2014.02.021⟩. ⟨pasteur-01420527⟩
HIV cure research: advances and prospects
Caroline P. Passaes a,b , Asier Saez-Cirion a
a Unité de Régulation des Infections Rétrovirales, Institut Pasteur, 25 –28 rue du Dr Roux, 75724 Paris Cedex 15, France.
b CEA, Division of Immuno-Virology, iMETI/DSV, 18 Route du Panorama, 92265 Fontenay-aux-Roses, France.
Email addresses:
caroline.pereira-bittencourt@pasteur.fr/caroline.passaes@cea.fr
asier.saez-cirion@pasteur.fr
Corresponding author:
Asier Saez-Cirion
Unité de Régulation des Infections Rétrovirales - Institut Pasteur
25 –28 rue du Dr Roux, 75724 Paris Cedex 15, France
Tel: +33 145688944; Fax: +33 145688957
E-mail: asier.saez-cirion@pasteur.fr
Abstract
Thirty years after the identification of HIV, a cure for HIV infection is still to be achieved. Advances of combined antiretroviral therapy (cART) in recent years have transformed HIV infection into a chronic disease when treatment is available. However, in spite of the favorable outcomes provided by the newer therapies, cART is not curative and patients are at risk of developing HIV-associated disorders. Moreover, universal access to antiretroviral treatment is restricted by financial obstacles. This review discusses the most recent strategies that have been developed in the search for an HIV cure and to improve life quality of people living with HIV.
Keywords:
HIV cure, eradication, remission of HIV infection, HIV reservoirs, reactivation of latent virus, repression of provirus, control of infection.
Highlights:
- Some cases of cure or remission of infection have boosted the search for an HIV cure
- cART intensification has not shown significant impact in the reservoirs, but early cART may limit them
- Strategies to purge the reservoirs face difficulties linked to the complexity of latency mechanisms and drug non-specificity
- Repression of reservoirs or cell manipulation to render them less permissive to HIV may facilitate HIV remission
- HIV cure/remission may require boosting immune responses while keeping inflammation in check
Preamble
The introduction of improved combined antiretroviral therapy (cART) has dramatically improved the clinical outcome and life expectancy of HIV infected patients. The development of drugs that inhibit different steps of viral replication allows clinicians to successfully manage the disease, improving immunologic parameters, and avoiding drug resistance and side effects. Due to advances in antiretroviral development, HIV infection can nowadays be handled as a complex chronic disease ( Deeks et al., 2013 ). Nonetheless, despite all of the clinical benefits provided by drug therapy, cART alone is not able to eradicate the virus, which persists in reservoirs that are thought to be the source for viral reemergence after treatment interruption ( Chun et al., 2010 ; Chun et al., 1997 ). In addition, cART does not fully restore health – it increases the risk of non-AIDS disorders such as cardiovascular, kidney, liver and neurological diseases. Moreover, persistent immune dysfunction and inflammation increase the risk of non-AIDS morbidity and mortality ( Clifford and Ances, 2013 ; Lake and Currier, 2013 ). Undoubtedly, a cure for HIV infection is needed to bypass the limitations of the current therapy and restore health. Although a vaccine against HIV remains an unachieved aspiration, several recent findings suggest that either a cure or a durable remission of infection might be possible.
Is a cure for HIV possible?
Since the isolation and characterization of the HIV as the etiologic agent of AIDS (Barre-Sinoussi et al., 1983 ), the search for a cure has been considered a major research priority . The description of a unique case of HIV cure after hematopoietic stem cell (HSC) transplantation has boosted remarkable optimism in the field and rekindled the hope that a cure for HIV infection is possible. In 2007, the so-called “Berlin patient” - HIV positive and diagnosed with acute myeloid leukemia - received double allogeneic HSC transplant from an HLA-matched and unrelated donor screened for homozygosity for the CCR5 ∆32 allele ( Hutter et al., 2009 ). This patient discontinued ART the day before the first transplantation and, after 6 years of follow up in the absence of therapy, he shows no trace of HIV in blood and tissue samples (Figure 1A), revealing no evidence for persistent HIV. Levels of HIV-specific antibodies have also declined, suggesting that HIV antigen stimulation was very low or absent after transplantation ( Allers et al., 2011 ; Hutter et al., 2009 ; Yukl et al., 2013 ).
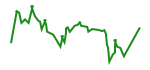
Several studies have aimed to use autologous or allogeneic HSC transplantation in association with antiretroviral therapy as a strategy to eradicate HIV in seropositive patients diagnosed with leukemia and/or lymphomas (Table 1). Nevertheless, in most of these studies HIV was detected after transplantation, either following therapy withdrawal or because the therapeutic regimen was not able to completely eliminate the viral reservoirs. In addition, in several cases, the patients died after transplantation. The most recent study investigating the impact of cART on viral reservoirs in two patients who received HSC transplantation – known as “the Boston patients” – described an important reduction of HIV DNA in long term, reaching undetectable levels ( Henrich et al., 2013 ). These observations are in agreement with another study which described that HIV DNA levels at month 24 post transplantation in HSC transplanted patients are lower than those observed at baseline ( Simonelli et al., 2010 ). Altogether, these data suggest that HSC transplantation in association with cART may reduce viral reservoirs in HIV infected patients. However, a recent report during the 6th International Workshop on HIV Persistence, Reservoirs and Eradication Strategies revealed that the Boston patients experienced a strong rebound in plasma viremia several months after analytic treatment interruption (ATI) (Figures 1B and 1C)( https://www.carmenthyssenmalaga.org ). These results suggest that HIV hides in yet unidentified sanctuaries or at levels that are not detectable by current available techniques.
So far, HSC transplantation to eradicate the virus was successful only in the Berlin patient. The mechanisms involved in HIV eradication in this patient are not yet fully understood. This patient underwent severe particular transplant conditions ( Hutter et al., 2009 ) (Figure 1), such as double HSC transplantation from the same CCR5 ∆32 donor, conditioning regimen (total body irradiation) and the development of graft-versus-host disease (GVHD), which may have contributed to cure. Comparison with the protocols applied in the case of the Boston patients ( Henrich et al., 2013 )suggests that total body irradiation or the engraftment with cells from a CCR5 ∆32 donor may have been critical differential aspects in the case of the Berlin patient. As the homozygous CCR5 ∆32 deletion offers a natural resistance to HIV infection (Dean et al., 1996 ), this unique successful experience has encouraged the search for new CCR5 -based therapeutic approaches for HIV cure interventions (see below). It is interesting to notice that the Berlin patient and both Boston patients were originally heterozygous for the CCR5 ∆32 allele, but this did not confer any advantage to the Boston patients once treatment was interrupted ( Henrich et al., 2013 ; Hutter et al., 2009 ). A recent effort to cure HIV infection in a 12-year old boy with acute lymphoblastic leukemia consisted of an allogenic transplantation of HSC obtained from cord blood. Similar to the Berlin patient, the donor was screened for homozygosity for the CCR5 ∆32 allele. This procedure was performed at the University of Minnesota in April 2013, but the pediatric patient died two months after transplantation by a severe GVHD (https://www.carmenthyssenmalaga.org ).
Despite the impact of HSC transplantation on reducing HIV reservoirs, this kind of treatment is not a viable option for the majority of HIV infected patients, since it is a risky and expensive procedure that is recommended only for those who develop cancer. Furthermore, CCR5 ∆32 homozygous donors are rare (1% of caucasians) (Lucotte, 2002 ), what makes the search for a compatible donor with the protective genotype an additional challenge.
How can HIV infected patients achieve durable remission of infection?
“HIV controllers” or “elite controllers” correspond to a small percentage of HIV infected patients that can naturally control viral replication below the levels of detection with standard clinical assays. Consequently, this group of patients is considered an important model to understand the mechanisms underlying control of infection in the absence of treatment ( Hamimi et al., 2013 ; Saez-Cirion and Pancino, 2013 ). The aim of a so-called “functional cure” would be similarly to HIV controllers, that is, to allow HIV infected patients to achieve viral remission in which HIV remains in the body at low levels and it is controlled by the host in the absence of cART. This status may be achieved by 5-15% of patients treated very early during primary HIV infection (PHI) for long periods of time who experience treatment interruption afterwards ( Goujard et al., 2012b ; Hocqueloux et al., 2010 ; Lisziewicz et al., 1999 ; Lodi et al., 2012 ; Saez-Cirion et al., 2013 ; Salgado et al., 2011 ; Steingrover et al., 2008 ). These patients are known as “post-treatment controllers”.
The VISCONTI study (Viro-Immunological Sustained CONtrol after Treatment Interruption) investigated whether the characteristics of 14 post-treatment controllers were similar to those observed in HIV controllers that spontaneously control HIV replication ( Saez-Cirion et al., 2013 ). In this study, cART duration at after primary infection was 3 years, and after treatment interruption, post-treatment controllers presented a sustained control for a median of 7 years. During acute phase, post-treatment controllers had higher viremia levels and lower CD4+ T cell counts than patients who naturally control infection afterwards. In addition, these groups of patients have a different genetic background – while HIV controllers cohorts are enriched by the protective HLA-B*27 and B*57 ( Migueles et al., 2000 ; Pereyra et al., 2008 ; Saez-Cirion et al., 2007 ), the risk alleles B*07 and B*35 ( Carrington et al., 1999 ; Itescu et al., 1992 ; Pereyra et al., 2010 ; Peterson et al., 2013b ) were prevalent among the 14 post-treatment controllers. HIV-1 specific CD8+ T cell responses and immune activation differ between post-treatment controllers and HIV controllers (post-treatment controllers have weak CD8+ T cell responses and a low level of immune activation during the control phase).
Another recent incident that suggests that a very early treatment may lead to HIV remission is the case identified as ‘the Mississipi baby” (Persaud et al., 2013 ). This infant born to a seropositive mother started receiving cART 30 hours after birth. HIV-1 RNA was detectable at 31 hours, days 6, 11 and 19, and reached undetectable levels at day 29. cART was discontinued sometime between month 18 to 23. At 36 months of age and after therapy withdrawal, HIV-1 RNA, proviral DNA and HIV-1 antibodies remain consistently undetectable or extremely low in blood and tissues in this pediatric patient.
The study of post-treatment controllers may give valuable information to guide the search for a successful strategy for inducing viral remission. However, some open questions remain. For instance: which are the mechanisms underlying control? and how can we increase the probability of HIV infected patients to become a post-treatment controller? Recent studies suggest that both the timing to initiate therapy (Ananworanich et al, 20th Conference on Retroviruses and Opportunistic Infections, 2013, San Francisco, USA) ( Ananworanich et al., 2012 ; Gianella et al., 2011 ; Hecht et al., 2006 ) and the duration of cART ( Fidler et al., 2013 ; Stohr et al., 2013 ) might play a key role in the achievement of a durable HIV control. In addition, baseline viral load and immune activation may predict success ( Volberding et al., 2009 ). Whether the therapeutic regimen used during PHI may impact the outcome is still to be investigated. In any case, in the absence of markers predicting success after treatment interruption, this procedure is not recommended outside structured protocols for patients undergoing suppressive cART.
Treatment during PHI seems to result in broad and strong HIV-1 specific immune responses ( Hecht et al., 2006 ; Kaufmann et al., 2004 ; Lisziewicz et al., 1999 ; Moir et al., 2010 ; Ortiz et al., 1999 ; Oxenius et al., 2000 ; Rosenberg et al., 2000 ), reduced immune activation (Volberding et al., 2009 ), immune restoration in the gastrointestinal mucosa (Guadalupe et al., 2006 ) and limited viral evolution (Chamberland et al., 2011 ). Additionally, initiation of cART during PHI may limit the establishment of viral reservoirs ( Archin et al., 2012b ; Chun et al., 2010 ; Gianella et al., 2011 ; Pires et al., 2004 ; Schmid et al., 2010 ; Wyl et al., 2011 ). Central memory CD4+ T cells (T CM ) are a key component of the long-lasting HIV reservoir ( Chomont et al., 2009 ), and recent studies have demonstrated that very early cART limits the seeding of the HIV reservoir in long-lived T CM (Figure 2) (Ananworanich et al, 20th Conference on Retroviruses and Opportunistic Infections, 2013, San Francisco, USA). Reservoirs levels in post-treatment controllers during the control phase were very low and it